Statin safety and efficacy hyped by flawed studies and false advertising
As documented in Parts 1 & 2, no studies had ever shown that dietary fat increased cholesterol or that lowering cholesterol prevented coronary atherosclerosis. This changed with the advent of statins, especially Lipitor (atorvastin), which reported a 36 % reduction in heart attacks by using relative risk statistics rather than actual risk of l %. Crestor was more potent than Lipitor but it was also associated with more adverse side effects. In March 2004, only eight months after its release, the Public Citizen consumer advocacy group asked the FDA to ban Crestor. Three cases of kidney failure associated with severe rhabdomyolysis (destruction of striated muscle) and one death had already occurred in the U.S. and seven cases of rhabdomyolysis and nine of kidney failure had been reported in Canada and the UK. That was ominous, as is well established the vast majority of adverse drug reactions are never reported. Baycol, an early statin, was approved by the FDA in 1997 and by the time it was banned in 2001, 1,899 cases of rhabdomyolysis and numerous deaths from kidney failure had been documented, many of which might have been prevented since they occurred long after unequivocal evidence that the drug should have been withdrawn. A 2005 study revealed that kidney failure and muscle weakness were two to eight times more frequent among Crestor users than those taking Lipitor and Zocor (simvastatin) [1].
Nevertheless, there allegedly were no unusual side effects or elevation of liver enzymes from the 40-mg. maximal dose of Crestor taken daily for two years in the ASTEROID trial. This was the first study to show a regression of plaque volume based on intracoronary ultrasound (IVUS) measurements after treatment, as shown below in Figure 1.
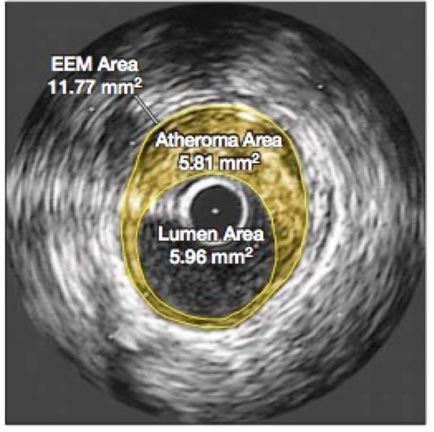
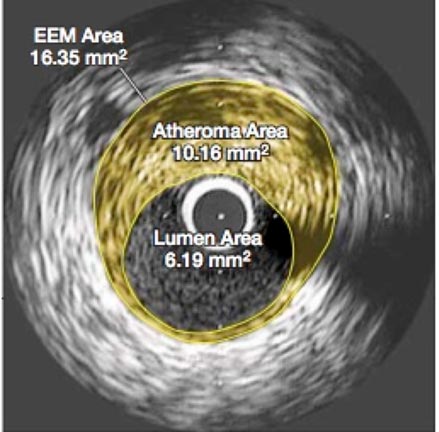
Figure 1 Effect of very high-intensity statin therapy on regression of coronary atherosclerosis in the ASTEROID trial; left before treatment, right after treatment [2]
It was the cross-sectional areas of atheroma that were compared before and after treatment, as it was assumed that these measurements were directly proportional to their volumes and that that the area of the lumen would show a corresponding increase. However, what was not discussed in either the press releases or the article was that the lumen area actually decreased by 4 %, and as can be seen, the images also showed that the arterial wall had thickened. This might not be beneficial because a smaller lumen and a stiffer arterial wall would both tend to increase blood pressure, an effect also not addressed in the published report. The Comments section conclusion said [2], “This very intensive statin regimen was well tolerated”. But the total dropout rate appears to have been 25 %; no details were provided to explain that. Also, the trial may have been too short for the Crestor side effects reported in other studies to have surfaced, raising questions about long-term safety with this large daily dose that would have to be taken perpetually. In addition, there were no controls and only 349 patients.
Crestor really made headlines with the subsequent JUPITER trial in men over 50 and women over 60 with no history of heart disease but who had an elevated C-Reactive Protein (CRP) and a normal LDL [3]. These 17 802 healthy people received either 20 mg of Crestor daily or a placebo and were followed to document the occurrence of the following end points: fatal and nonfatal myocardial infarction, fatal and nonfatal stroke, arterial revascularization, hospitalization for unstable angina or death due to a confirmed cardiovascular cause. It was scheduled to run until 520 end points had been reached. However, the way end point statistics were collected, one person might be recorded as having several, even though they had only one incident or hospitalization. There was so much overlap that it is difficult to know exactly what was being reported. It was anticipated that the study might last four or five years, but it was stopped after 23 months at which time only 393 end points had been reported. Although an “unequivocal reduction in cardiovascular mortality” was publicly announced as the major reason, the data did not support that. The only justification for termination was that the placebo group had experienced 109 more of these confusing end points and it was felt that continuing the study could subject them to increased harm.
This was exaggerated in reports claiming that Crestor reduced by almost 50% the risk for a major first cardiovascular event; but this was relative risk (RR) As can be seen below, the actual risk reduction (AR) was less than 1%.
Rate of primary endpoint: Crestor 1.6%; placebo 2.8% → AR, 1.2%.
Rate of fatal or nonfatal MI: Crestor 0.35%; placebo 0.76% → AR 0.41%.
Rate of fatal or nonfatal stroke: Crestor 0.37%; placebo 0.72% → AR 0.35%
It would appear from the above that the Crestor group indeed had slightly less than half as many heart attacks, but they also had 150 % more fatal heart attacks. The data was presented in a manner that obscured this, as follows:
Any myocardial infarction: Crestor 31, Placebo 68
Nonfatal myocardial infarction: Crestor 22, Placebo 62
To find the number of fatal heart attacks, subtract "Nonfatal myocardial infarction" from "Any myocardial infarction". This reveals 9 deaths in those receiving Crestor (29 %), compared to 6 in the placebo group (9%). Stroke was similarly presented to show a 50 % reduction from Crestor:
Any stroke: Crestor 33, Placebo 64
Nonfatal stroke: Crestor 30, Placebo 58
This means there were 3 fatal strokes in the Crestor cohort and 6 in those taking a placebo, so that total cardiovascular deaths (12) were identical in both groups.
Conflicts of Interest, sponsor control, & problems with the placebo group and diabetes
There were also concerns about conflicts of interest and the role of the sponsor. The lead author is a co-holder of the patent for the hsCRP test used, which became the standard method of measurement at $50.00/test. Nine of the 14 authors had significant financial ties to AstraZeneca, whose investigators also collected, controlled and managed the raw data and monitored the collection sites. It is well established from other drug company sponsored studies that bias can creep in, such as the preponderance in the placebo group of patients with a family history of heart disease or metabolic syndrome, both of which significantly increase risk. Crestor is a potent statin with numerous side effects, but in JUPITER, there were just as many side effects in the placebo group. The most common adverse side effect is musculoskeletal pain, which occurs in 25 % [4]. But only 19 out of the 18 000 subjects taking part in the trial reported this symptom, 10 in the Crestor group and 9 in the placebo group. At the time the study was terminated, one out of four were not taking their medication. No reason was given for this nor do we know why or how many of the deaths came from this group. The fact is that there was a difference of less than 50 deaths between the two groups during the study, which given the large number of participants, means that neither group was at a significantly increased risk of dying.
Read more
Read more
No comments:
Post a Comment